 Filling up the different orbitals by the number of electrons will follow these lines configuration.Now inclined parallel lines are drawn through the electronic orbitals according to the above picture.The different electron orbitals originating from the same electronic energy levels are written in horizontal lines.Therefore, the trivial way but most convenient way to remember these electronic energy levels is given below the diagram. The hydrogen atom contains only one electron in 1s hydrogen energy levels with electronic configuration 1s 1.īut difficult for readers to remember the electron energy levels diagram for many electronic configurations. The energy associated with a certain energy level increases with the increase of its distance from the nucleus. What are electron energy levels?Įnergy levels are fixed distances where electrons are rotating around the nucleus with definite energy. The modern periodic table classification like s, p, d, and f block elements is based on properties and general electron or electronic configuration of elements. For example, 3s orbital has lower energy than 3p orbitals which again lower energy than the 3d level. To find the electron configuration formula first we find the order of electronic energy levels of s, p, d, and f orbitals or sub-levels. Hence the organic and inorganic chemical reactions are better understood by the electronic configuration of elements. So those are the exceptions the main exceptions that you'll see when dealing with lifetime configuration.Oxidizing reducing properties, oxidation number, ionization energy, electron affinity, shielding effect, the polarity of chemical bonds, acids bases properties can be explained by the electronic configuration.Ī reaction to reach chemical equilibrium is the change of electron configuration of reactant and product atoms.  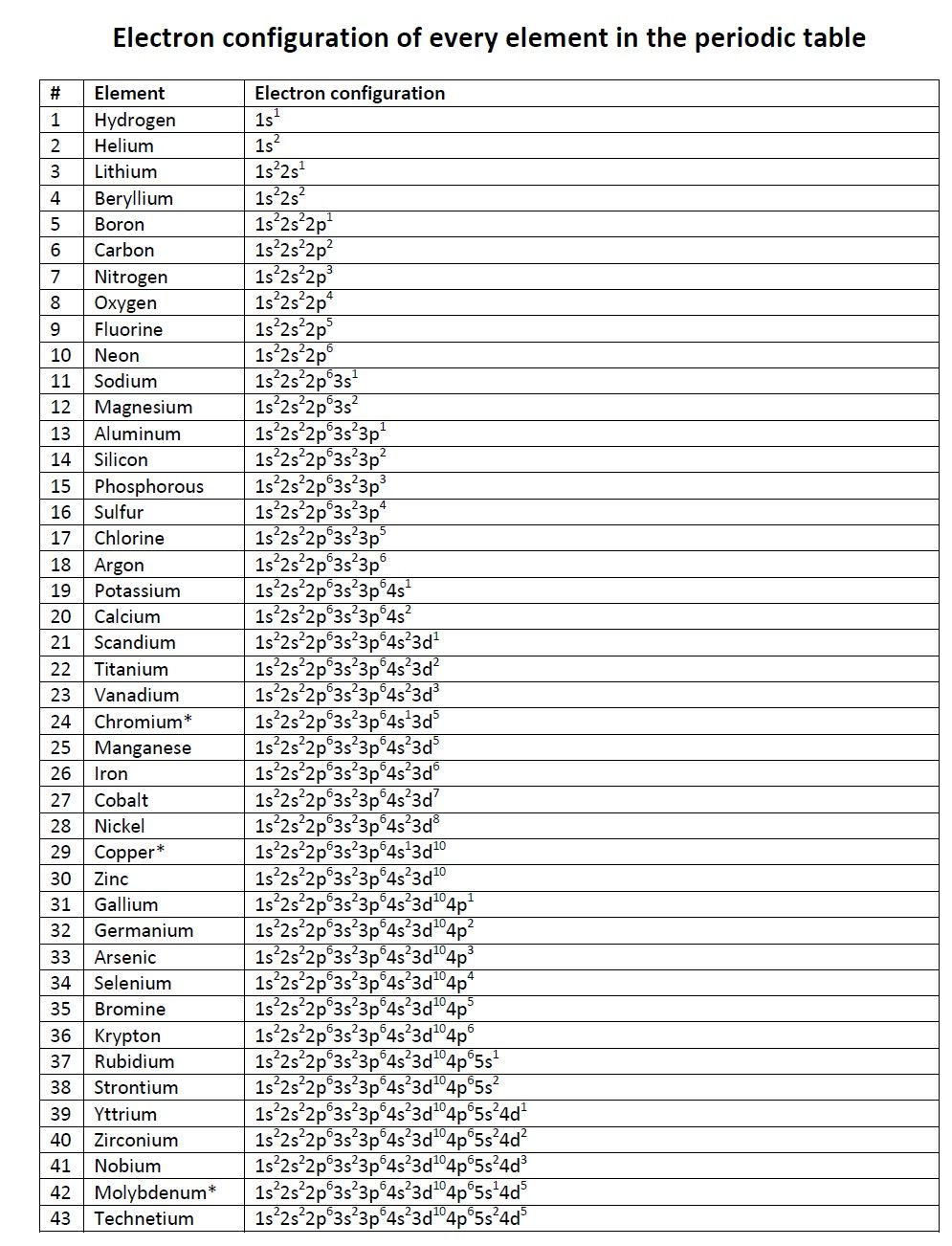
So, again, I'm going to make this argon just make it for myself 4s1, 3d10 and again you might see it as argon 3d10, 4s1 same exact thing. That way this d orbital or d sublevel be completely filled which is very stable versus the s orbital will be halfway filled. Okay so anytime that it ends in d9 we're going to fix it up a little bit, same exact way we're going to take an electron from the 4s and we're going to move it over to the 3d. So let's make the electron configuration for that 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d9 from here on out anytime you see d9 we're going to change it. So you might see either one but they're the same.ĭown here the other exception that you're going to see is copper, or anything along copper which we'll talk about. So this is talking about the order of energy you might see it also looking like this, this is just doing it in order of number 3 comes before 4 but they're exactly the same thing, they're depicting the exact same thing, nothing different about them. 
You might see this is the diagram or the electron configuration that you're going to see and this is actually higher in energy than this. So instead we're going to write actually we're going to make this argon as we noted before in the noble gas configuration and we're going to make this 4s1, 3d5 this is halfway filled which is pretty stable and this is much more stable being half way filled rather than being the d4. So what we're going to do, we're going to, that should be a 2 there, sorry, and we're going to take one of the electrons in the 4s orbital and move it over to the 3d orbital. Right now we're 1 electron short of it being halfway filled. 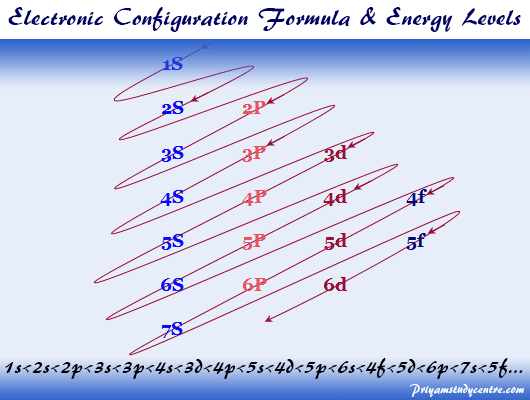
The d sublevel is more stable when its either half full all the orbitals are filled with at least 1 electron or completely filled. If we're going to make this short hand and make the electron configuration for this we would make this 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d4 okay from now on every time you see 3d4 you're going to change it, we do not like 3d4. Chromium is a transition metal and it has 24 electrons and here is the orbital diagram. Okay alright so let's talk about the exceptions you're going to see when you're dealing with electron configuration there's going to be a few around but we're going to talk about the main ones you're probably going to see in class. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
